On the Origins of New Forms of Life
4.4: Polyploid Production
(Continued from the previous page)
 Bread Wheat, Triticum aestivum (lower panel), and
the two parents that produced it, rivet wheat, T. turgidum (upper left) and Tausch's goatgrass, Aegilops tauschii (upper right).
Credits: Bluemoose
(upper left) and Stan Shebs (lower panel)
Bread Wheat, Triticum aestivum (lower panel), and
the two parents that produced it, rivet wheat, T. turgidum (upper left) and Tausch's goatgrass, Aegilops tauschii (upper right).
Credits: Bluemoose
(upper left) and Stan Shebs (lower panel)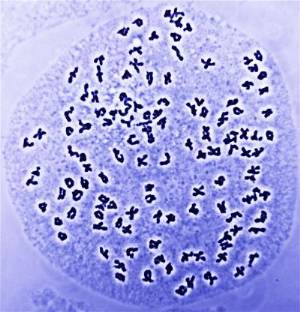 Polyploidy is important in the origin of new forms of life and sometimes produces genomes with a large number of chromosomes. This photograph shows a chromosome squash of a hexaploid fern of the genus Cystopteris with 126 pairs of chromosomes. Credit: Michael Windham.
Polyploidy is important in the origin of new forms of life and sometimes produces genomes with a large number of chromosomes. This photograph shows a chromosome squash of a hexaploid fern of the genus Cystopteris with 126 pairs of chromosomes. Credit: Michael Windham. Oilseed rape (Brassica napus), a common polyploid crop plant that was produced by the hybridization of Brassica oleracea and Brassica rapa. Credit: John Innes Centre
Oilseed rape (Brassica napus), a common polyploid crop plant that was produced by the hybridization of Brassica oleracea and Brassica rapa. Credit: John Innes Centre
Polyploid organisms have three or more complete sets of chromosomes. As might be expected, they are produced by processes that duplicate chromosome sets. They arise from preexisting organisms via the addition of one or more entire extra sets of chromosomes. Diploid organisms have two sets of chromosomes. Haploid organisms have only one.
Polyploids with a particular number of chromosome sets are designated by names that reflect their level of ploidy (triploids have three sets, tetraploids have four, pentaploids have five, and so forth). Each type of polyploid organism is a distinct chromotype and each corresponding population is a distinct chromoset. Any process giving rise to polyploids is known as polyploidization. Polyploidization is a kind of chromosomal mutation. Polyploids are of two basic types. Allopolyploids are polyploids that arise through hybridization, while an autopolyploid is a polyploid that arises from a single parental form. Beetleweed (Galax urceolata) is an example of a natural autopolyploid.1 Actually, beetleweed has both a diploid and tetraploid form, which are treated as conspecific. The two are similar except that the tetraploid is substantially larger, and a bit sturdier, with thicker leaves. Their distribution in eastern North America is also similar, with the tetraploid ranging somewhat wider.2
In the case of an allopolyploid, chromosome doubling stabilizes the reproductive cycle because it confers fertility. Hybrids produced by interbreeding between two diploid organisms normally receive one haploid set of chromosomes from each of their parents. Thus, as has already been mentioned, hybrids derived from matings between distinct chromotypes will have some or all of their chromosomes unpaired. The result is structural heterozygosity, which has a severe adverse effect on the fertility of the hybrids. However, when all of a hybrid's chromosomes are doubled, something that commonly occurs, all are paired once again. The resulting allopolyploid is fully fertile.3 An allopolyploid that has achieved fertility by such means is known as an amphiploid. Amphiploids are of interest in an evolutionary context because they often have adaptively superior combinations of parental characters.4
The simplest case is the situation just described of an amphiploid derived from matings between two diploid parents. However, in any type of hybrid, chromosome doubling eliminates any sterility due to mismatched chromosomes. For example, a tetraploid might hybridize with a diploid. The offspring would therefore normally be triploid. Chromosome pairing is always poor in triploids (or in any odd-number polyploid) so fertility is correspondingly low. However, in the example just given, if chromosome doubling occurred, the resulting fertile hexaploid would have all chromosomes paired. Obviously, doubling the chromosomes will always produce a fully paired karyotype, no matter what kind of hybrid is in question.
Common bread wheat (Triticum aestivum) is an example of a common amphiploid. This hexaploid came into being about 7,000 years ago from hybridization between rivet wheat (T. turgidum), a tetraploid, and Tausch's goatgrass (Aegilops tauschii), a diploid.5 In fact, Talbert et al. (1998) say it arose repeatedly as the result of multiple hybridization events. Rivet wheat itself is a much older, natural amphiploid derived from hybridization between two diploid grasses. One was the goatgrass Aegilops speltoides.6 The other was either wild einkorn (Triticum boeoticum) or another wild wheat, T. urartu, more likely the former.7 Another natural amphiploid is Plagiomnium medium (medium plagiomnium moss), shown by Wyatt et al. (1988) to be derived from hybridization between P. ellipticum (elliptic plagiomnium moss) and P. insigne (plagiomnium moss).
Specific Modes of Polyploid Production. Polyploids arise in various ways. Here we will consider two such processes that are actually known to occur (others have been proposed, but appear to be poorly documented):
- unreduced gametes;
- somatic multiplication.
The latter of these two modes of polyploidization seems to be the more common, but for those readers who are interested the relative prevalence of these two main ways in which polyploids arise is discussed in Appendix B. Relevant evidence is discussed at some length in that appendix, but in short, well-established facts indicate that polyploid production via unreduced gametes is extremely common in a natural setting. Somatic doubling may also be a common natural occurrence, but more research is needed to establish this. A third type, zygotic doubling (see Appendix C), has been observed many times under experimental conditions, but it is unclear how often it occurs under natural circumstances.
Unreduced Gametes. The terms haploid, diploid, triploid, and so forth, are applied also to gametes (and not only to the organisms that produce those gametes). Thus, a gamete that contains one set of chromosomes is haploid. One that contains two is diploid, and so forth. Therefore, when a haploid gamete (with one set of chromosomes) fertilizes a diploid gamete (with two sets of chromosomes), the resulting organism is triploid (it has three sets of chromosomes). A second hypothetical example would be the fertilization of a triploid egg by a diploid sperm, which would produce a pentaploid offspring.
The usual product of meiosis is a gamete containing half as many chromosome sets as do the cells of the organism producing the gamete. For example, a diploid organism normally produces haploid sperm. Such gametes are called "reduced" because the number of chromosome sets they contain has been reduced relative to the number present in the organism producing them. Therefore, when two such haploid gametes fuse during fertilization, a diploid offspring with two sets of chromosomes is produced (Figure 4.2a). This process produces a stable life cycle where parents produce offspring with the same number of chromosomes as themselves: The diploid phase of the organism produces haploid gametes; haploid gametes fuse to produce diploid phase, and so forth.
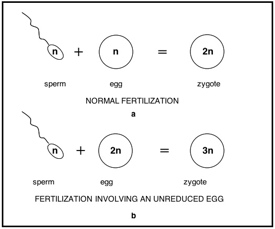 Figure 4.2 (a) Two haploid gametes fuse to produce a
diploid zygote; (b) A haploid gamete and a diploid
gamete fuse to produce a triploid zygote.
Figure 4.2 (a) Two haploid gametes fuse to produce a
diploid zygote; (b) A haploid gamete and a diploid
gamete fuse to produce a triploid zygote. Brittlestem Hempnettle
Brittlestem HempnettleGaleopsis tetrahit
However, diploid organisms also produce some diploid gametes along with their normal, haploid gametes.8 Such gametes are called "unreduced" (there has been no reduction in chromosome number versus the producing organism). Diploid organisms produce unreduced gametes that are diploid, but polyploids can produce unreduced gametes that contain more than two sets of chromosomes. When unreduced gametes fuse, or when an unreduced gamete fuses with a haploid gamete, a polyploid offspring results (Figure 4.2b). This process produces organisms with chromosome counts differing from their parents'. For example, hybrids between maize (Zea mays) and eastern gamagrass (Tripsacum dactyloides) are male sterile, but female fertile.9 In the hybrids, reproductively functional eggs are diploid.10 In backcrosses these eggs fuse with haploid pollen from either parent to produce triploid offspring.11
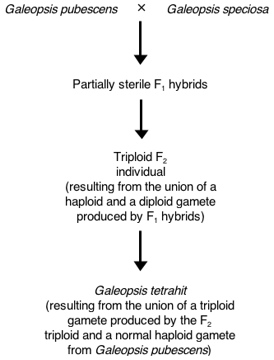 Figure 4.3: The three-step stabilization process
producing Brittlestem Hempnettle Galeopsis tetrahit
Figure 4.3: The three-step stabilization process
producing Brittlestem Hempnettle Galeopsis tetrahit
Polyploid organisms regularly produce gametes that would be considered unreduced if they were produced by a diploid organism.12 For example, a tetraploid normally produces diploid gametes. Chourrout et al. (1986) exploited this fact to produce a triploid breed of rainbow trout (Oncorhynchus mykiss) by crossing diploid and tetraploid forms. The haploid gametes of the former united with the diploid gametes of the latter to produce triploid offspring. A triploid form of the loach Misgurnus anguillicaudatus (oriental weatherfish) spawns eggs containing one, two, or three sets of chromosomes.13 Matsubara et al. (1995) fertilized triploid eggs from this source with haploid sperm to produce tetraploid offspring.
Müntzing (1930, 1932) proved the tetraploid annual herb Galeopsis tetrahit (brittlestem hempnettle) is derived from hybridization. G. tetrahit has been treated as a species since the time of Linnaeus. Müntzing synthesized it via a three-step process (diagrammed in Figure 4.3). He first crossed Galeopsis pubescens (Downy Hempnettle) and G. speciosa (Large-flowered Hempnettle). The resultant hybrids were quite infertile and exhibited irregular meiosis. They had only about eight percent viable pollen. The F₂ generation was composed mostly of diploids, but there was one triploid that arose from the union of a haploid gamete and a diploid gamete.14 When Müntzing backcrossed this triploid individual to G. pubescens, it produced a single seed, which grew into a tetraploid plant. This tetraploid individual arose when a triploid egg was fertilized by a normal haploid gamete from G. pubescens.15 It produced 70 percent viable pollen and yielded fertile progeny via self-fertilization. These artificial allopolyploids matched G. tetrahit in morphology and karyotype. Moreover, they produced fertile progeny when crossed with G. tetrahit, but were reproductively incompatible with G. pubescens and G. speciosa. Therefore, naturally occurring G. tetrahit almost certainly arose via a similar process.
Another case of a new stable type produced by the union of unreduced gametes is the tetraploid hybrid Karpechenko (1927, 1928) obtained by crossing cabbage and radish (Brassica oleracea × Raphanus sativus). In this cross, the F₁ hybrid is quite sterile due to a nearly complete lack of chromosome pairing. However, it does produce some viable diploid gametes. Fusion of such gametes gave rise to Karpechenko's tetraploid, the "rabbage" known as radicole (Raphanobrassica), in the F₂ generation. These F₂ plants exhibited regular meiosis and were fertile. Moreover, they were reproductively stable, yielding later generations morphologically similar to the F2.16
Pinkava et al. (1985: 473) say that a diploid chromotype of the cactus Cylindropuntia fulgida (jumping cholla) provided both a diploid and a haploid gamete that joined to produce an autotriploid chromoset (treated as conspecific with C. fulgida). Like most triploids, these autotriploids are quite sterile. However, they propagate themselves clonally via efficient vegetative reproduction (through easily detached stem segments).
 Blue-spotted Salamander
Blue-spotted SalamanderAmbystoma laterale
Uzzell (1963, 1964) reported that two North American salamanders, Ambystoma platineum (Silvery Salamander) and A. tremblayi (Tremblay's salamander), are triploid parthenogens. Sessions (1982) showed they had both been produced via a two-step stabilization process: (1) an A. laterale (Blue-spotted Salamander) male crossed with an A. jeffersonianum (Jefferson Salamander) female to produce a female hybrid; (2) this female hybrid produced diploid eggs that were fertilized by A. jeffersonianum males, which produced A. platineum. Fertilization of the same eggs by A. laterale males produces Both of these salamanders are parthenogens.
When multiple spermatozoa fertilize a single egg, the effect on the chromosome number of the fertilized egg (i.e., on the zygote) can be the same as fertilization by a single unreduced male gamete. However, while the production of polyploids by this method has in fact been observed,17 it seems much rarer than polyploidy induced by unreduced gametes, presumably because the eggs of most organisms have a mechanism that efficiently prevents the entry of multiple spermatozoa.
Somatic Multiplication. A new polyploid form can also be produced when (1) a chromosomal mutation increasing the number of chromosome sets occurs in a somatic cell (i.e., a cell that is not, and is not destined to become, a gamete); and (2) that cell goes on to form by cell division an entire polyploid segment within an organism that otherwise remains diploid; (3) that segment, or a portion of that segment, separates from the parent organism and/or produces offspring to found a new polyploid line. For example, the Kew Primrose (Primula kewensis) was produced via a stabilization process of this kind. Its origin was well documented.18 First, an F1 hybrid was produced between the primroses P. floribunda and P. verticillata. It was diploid and highly sterile, but was propagated via cuttings. Later, in three separate years, this otherwise sterile clone produced fertile tetraploid shoots that gave rise immediately to the fertile, constant tetraploid now known as P. kewensis (these shoots each developed on the diploid hybrid from single cells in which chromosome doubling had occurred). In this way, the Kew Primrose emerged as a new type of plant with a stable sexual reproductive cycle.19
Processes producing polyploids, such as those just described can generate whole assemblages of related polyploids ("polyploid complexes"). For example, a diploid with the chromosome set A, could cross with a second diploid having the complement B, to produce a tetraploid with the set AB. The second of the diploids could cross with a third, having the set C, to produce the tetraploid BC. Another cross (A × BC) could produce a hexaploid with the complement ABC. The continued production of polyploids in this manner can produce an array of forms with even higher levels of ploidy. Grant (1981) gives many examples of naturally occurring polyploid complexes. NEXT PAGE >>
1. Baldwin (1941); Stebbins (1947, 1950: 316).
2. Stebbins (1950).
3. Stebbins (1950: 222).
4. Soltis and Soltis (1993).
5. Dvorak et al. (1998).
6. Zhang et al. (2002).
7. Zhang et al. (2002).
8. DeWet (1980); Fankhausen (1945: 35–36); Ramsey and Schemske (1998).
9. Manglesdorf and Reeves (1939); Newell and deWet (1973).
10. DeWet and Harlan (1974).
11. DeWet and Harlan (1974).
12. Stebbins (1950); Pandian and Koteeswaran (1998).
13. Pandian and Koteeswaran (1998); Similarly, Bogart (2003) notes that triploid females in the Salamander genus Ambystoma produce both reduced and unreduced eggs.
14. Grant (1971: 192); Müntzing (1930, 1932).
15. Grant (1971: 192). The production of triploid eggs by the triploid intermediate in this case is not implausible given that Pandian and Koteeswaran (1998) report that a triploid form of the fish Misgurnus anguillicaudatus "simultaneously spawns small, intermediate and large eggs carrying n, 2n and 3n genomes."
16. Karpechenko wanted to develop a hybrid with the leaves of a cabbage and the root of a radish, but Raphanobrassica turned out to agriculturally useless because it has the root of a cabbage and the leaves of a radish. A much more successful related allopolyploid is the raparadish, an allopolyploid hybrid between Raphanus sativus and Brassica rapa (field mustard), which is widely grown as a fodder crop (Lange et al. 1989).
17. Pandian and Koteeswaran (1998); Ueda et al. (1986); Vigfusson (1970).
18. Digby (1912); Pellew and Durham (1916); Newton and Pellew (1929); Upcott (1939).
19. Hiesey et al. (1971) report a similar case in monkey flowers (Mimulus), where a diploid F1 hybrid produced a tetraploid shoot.
Most shared on Macroevolution.net:
Human Origins: Are we hybrids?
On the Origins of New Forms of Life
Mammalian Hybrids
Cat-rabbit Hybrids: Fact or fiction?
Famous Biologists
Dog-cow Hybrids
Georges Cuvier: A Biography
Prothero: A Rebuttal
Branches of Biology
Dog-fox Hybrids